Table of Contents >> Show >> Hide
- Why “Identifying an Element” Is Easier Than It Sounds
- 1) Use the Atomic Number: The Element’s Unbeatable Fingerprint
- 2) Decode the Chemical Symbol (and Avoid the “Co vs CO” Trap)
- 3) Use the Element’s Address: Group (Column) + Period (Row)
- 4) Check the Atomic Mass: The “Weighted Average” Clue
- 5) Use Electron Clues: Valence Electrons, Blocks, and Trends
- A Quick “Element Identification” Checklist (Use This on Tests)
- Common Mistakes (So You Can Avoid Them Like a Pro)
- Conclusion: The Periodic Table Is a Map, Not a Memorization Punishment
- Experiences That Make These 5 Methods “Click” (500+ Words)
The periodic table looks like a grid of tiny, color-coded secretslike a spreadsheet that went to chemistry camp and came back with a personality.
But here’s the plot twist: each little box is basically an element’s ID card. Once you know what to look for, you can identify any element
quicklywhether you’re doing homework, prepping for a quiz, or trying to figure out what “Fe” has to do with your cereal’s iron content.
In this guide, you’ll learn five simple, reliable ways to identify an element on the periodic table using clues that are already printed right there:
atomic number, symbol, location (group and period), atomic mass, and electron information. We’ll keep it practical, a little funny, and very “you can do this.”
Why “Identifying an Element” Is Easier Than It Sounds
When people say “identify an element,” they usually mean one (or more) of these tasks:
- Match an element’s name to its symbol (and vice versa).
- Find an element using its atomic number.
- Predict an element’s general behavior (metal? nonmetal? reactive?) by where it sits.
- Use the element’s atomic mass or electron configuration to confirm you’ve got the right one.
The trick is knowing which clue is the “license plate” (unique identifier) and which clues are “car features” (helpful confirmation).
Spoiler: the atomic number is the license plate.
1) Use the Atomic Number: The Element’s Unbeatable Fingerprint
What the atomic number tells you
The atomic number is the number of protons in an atom’s nucleus. That count is what makes an element an element.
Change the number of protons and you don’t get a “slightly different version”you get a completely different element.
How to identify an element by atomic number
- Find the atomic number on the element’s tile (it’s usually the smallest whole number on the box).
- Match it to the table’s sequence. The periodic table is organized by increasing atomic number from left to right.
- Read the symbol and name on that tile to identify the element.
Examples you’ll actually use
- Atomic number 8 → Oxygen (symbol O). Useful for understanding why oxygen bonds the way it does.
- Atomic number 17 → Chlorine (symbol Cl). Yes, the stuff in pools has a very organized address.
- Atomic number 26 → Iron (symbol Fe). “Fe” looks odd until you realize symbols sometimes come from older names.
If you only memorize one thing for quick element identification, memorize this:
Atomic number = identity. Everything else is supporting evidence.
2) Decode the Chemical Symbol (and Avoid the “Co vs CO” Trap)
Symbols are short, but picky
Every element has a unique chemical symbol, typically one or two letters (occasionally three for temporary names historically).
The capitalization matters:
- Co = Cobalt (an element)
- CO = carbon monoxide (a compound, and not a cute one)
When you’re identifying an element, treat capitalization like it’s the difference between “US” and “us.” One is a country, the other is a vibe.
Why some symbols look “random”
A lot of symbols come from older or Latin-based names:
- Na = Sodium (from “natrium”)
- Fe = Iron (from “ferrum”)
- Pb = Lead (from “plumbum”)
You don’t need to speak Latin to identify elements, but it helps to know that the symbol isn’t always a nicknameit’s sometimes a historical artifact.
Fast symbol-to-element identification method
- Read the symbol carefully (case-sensitive).
- Scan the table for that exact symbol.
- Confirm with the atomic number and the element name on the same tile.
Practical tip: if you’re working from a worksheet and the symbol seems “off,” check your handwriting.
A sloppy “S” and a sloppy “Cl” can lead to wildly different outcomes (like a lab partner’s eyebrow disappearing… hypothetically).
3) Use the Element’s Address: Group (Column) + Period (Row)
Groups and periods: what they mean
The periodic table is arranged in:
- Periods (rows): elements with increasing atomic number across the row.
- Groups (columns): elements with similar outer-electron patterns and often similar chemical behavior.
This is why elements in the same column often “act related,” like cousins who share the same laugh and the same impulsive decisions at family gatherings.
How location helps you identify an element
Location won’t replace atomic number for identification, but it’s excellent for quickly narrowing your options and predicting the element’s “type.”
Common families worth knowing
- Group 1: Alkali metals (very reactive; love making +1 ions)
- Group 2: Alkaline earth metals
- Groups 3–12: Transition metals (lots of useful metals live here)
- Group 17: Halogens (reactive nonmetals; often form −1 ions)
- Group 18: Noble gases (famously unbothered; low reactivity)
Example: identifying by “family resemblance”
Suppose you’re told an element is a halogen and sits in Period 3. Halogens are in Group 17, and Period 3 is the third row.
The element at that intersection is chlorine (Cl). You can confirm by checking its atomic number (17).
This approach is especially helpful on tests where you’re given partial information like “Group 1, Period 4” (that points you to potassium, K).
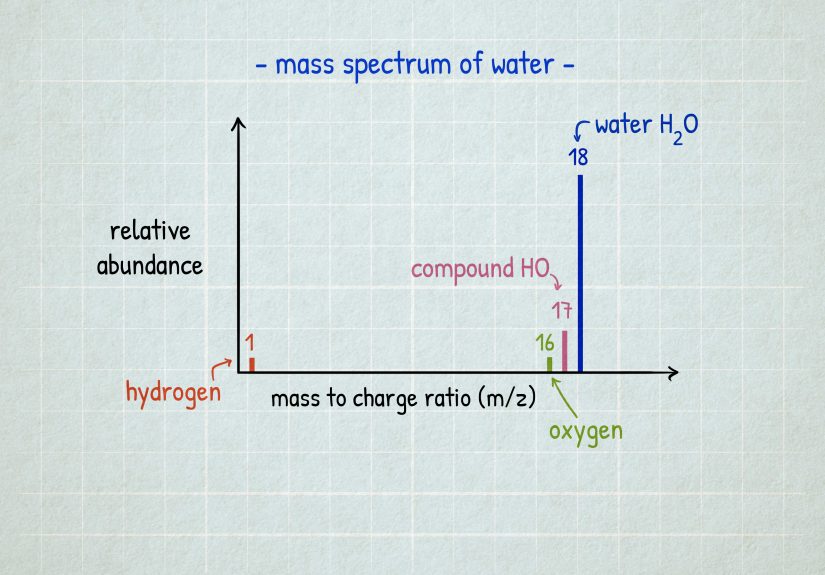
4) Check the Atomic Mass: The “Weighted Average” Clue
Atomic mass vs. mass number
The number on the element tile that looks like a decimal (for example, 35.45 for chlorine) is typically the element’s
standard atomic weight (often casually called “atomic mass” on many classroom periodic tables).
It’s a weighted average based on naturally occurring isotopes.
Meanwhile, a mass number is a whole number (protons + neutrons) for a specific isotope.
That’s why your periodic table shows decimals: nature mixes isotopes like a DJ mixes tracks.
How atomic mass helps identify an element
Atomic mass is best as a confirmation tool. If you think you’ve found the element, the atomic mass should be in the expected ballpark.
This is handy when:
- You’re distinguishing between elements with similar-sounding names.
- You’re verifying a symbol you might have copied incorrectly.
- You’re solving problems involving neutrons and isotopes.
A quick neutron estimate (with a reality check)
Many intro problems ask you to estimate neutrons by rounding the atomic mass to the nearest whole number and subtracting the atomic number:
- Example: Sodium (Na) has atomic number 11 and atomic mass about 22.99 (≈ 23).
- Estimated neutrons ≈ 23 − 11 = 12 neutrons (for the most common isotope).
Important: this is an estimate for common classroom work. Real isotope distributions and precise values can vary, and advanced chemistry
uses more exact isotope data. But for “identify the element” tasks, it’s a very practical cross-check.
5) Use Electron Clues: Valence Electrons, Blocks, and Trends
Valence electrons: the behavior drivers
Elements in the same group often share the same number of valence electrons (outer-shell electrons),
which heavily influences bonding and ion formation. That’s why group position can help you identify not just what an element is,
but how it tends to behave.
Block letters (s, p, d, f): a shortcut to electron structure
The periodic table is also divided into blocks based on where the outer electrons go:
- s-block: left two columns (plus helium) many reactive metals live here
- p-block: right side includes nonmetals, metalloids, and some metals
- d-block: middle transition metals
- f-block: bottom rows lanthanides and actinides
Using periodic trends to confirm an element
Periodic trends help you sanity-check an element’s identity and expected properties:
- Atomic radius: generally decreases left→right across a period, increases down a group.
- Ionization energy: generally increases left→right, decreases down a group.
- Electronegativity: generally increases toward the top-right (excluding noble gases).
For example, if you’re deciding whether an element is more likely to form a positive ion (metal behavior) or grab electrons (nonmetal behavior),
its position can guide you fast. Sodium (Na) on the left is famously eager to lose an electron; chlorine (Cl) on the right is eager to gain one.
Put them together and you get table salt (NaCl)which is chemistry’s way of saying opposites attract, and also your fries are now delicious.
A Quick “Element Identification” Checklist (Use This on Tests)
If you want a simple routine, here’s a reliable order:
- Atomic number: confirm identity first.
- Symbol + name: make sure you’re reading it correctly (capitalization matters).
- Group + period: verify you’re in the right neighborhood and predict basic behavior.
- Atomic mass: confirm the tile matches what you expect.
- Electron clues: use trends and valence patterns to double-check.
This works whether you’re identifying elements on a classroom chart, a digital periodic table, or a slightly wrinkled handout that looks like it survived a backpack hurricane.
Common Mistakes (So You Can Avoid Them Like a Pro)
- Mixing up symbols: “S” (sulfur) is not “Si” (silicon). One mistake and your “volcano experiment” becomes a “sandcastle experiment.”
- Ignoring capitalization: “Co” ≠ “CO.” One is cobalt; the other is a molecule that emergency rooms would prefer you not meet.
- Assuming atomic mass is a whole number: that decimal is normal and meaningful because isotopes exist.
- Forgetting the table’s legend: colors and labels vary. Some tables show states of matter, oxidation states, or isotope ranges.
Conclusion: The Periodic Table Is a Map, Not a Memorization Punishment
Identifying an element on the periodic table doesn’t require superpowersjust a consistent method. Start with the atomic number (the unbreakable ID),
read the symbol carefully, use group and period as your location clues, and confirm with atomic mass and electron-based patterns.
Once you practice this a few times, the periodic table stops feeling like a wall of random trivia and starts feeling like what it really is:
an incredibly organized cheat sheet made by scientists who love patterns almost as much as they love arguing about them.
Experiences That Make These 5 Methods “Click” (500+ Words)
Most people don’t fall in love with the periodic table the first time they see it. The usual first impression is closer to:
“Why are there so many boxes, and why does helium look like it’s hanging out with the noble gases even though it’s over there?”
Then something happensusually in a classroom, a lab, or a late-night study session with a snack you didn’t plan to eatand the table starts to make sense.
A common “aha” moment comes when you realize the atomic number is the boss of the whole operation. Students often try to identify elements by
memorizing names or matching colors, and that works… until it doesn’t. But when you use the atomic number, it’s like switching from guessing someone’s
identity by their hoodie to checking their driver’s license. Suddenly, you can stop arguing with yourself about whether you found sulfur or silicon and
just confirm it in seconds.
Another classic experience: the symbol trap. People who are otherwise calm and responsible have been known to write “AL” instead of “Al” and then wonder
why their answer doesn’t match the key. Once you’ve been burned by capitalization, you never forget it. It’s the chemistry equivalent of learning that
autocorrect is not your friend. After a few rounds of “Co vs CO,” you start reading symbols like a proofreader reading a contract.
Then there’s the “family resemblance” phasewhen you stop seeing the table as 118 separate facts and start seeing it as a set of neighborhoods.
This is where groups and periods feel like cheat codes. If you’ve ever noticed that lithium, sodium, and potassium are all described as very reactive
metals, you’ve basically met Group 1’s personality. That group-and-period method often shows up in real study habits: learners will cover the element
names with a sheet of paper and practice identifying what belongs where based on column behavior. It’s surprisingly satisfyinglike doing a puzzle where
the picture gradually appears.
Atomic mass becomes meaningful when isotopes enter the chat. The first time someone learns that chlorine’s mass is a decimal because it’s an average,
you can almost see the mental lightbulb flick on. It stops being “that annoying number under the symbol” and becomes evidence. Students often remember
this best when they do a problem estimating neutrons and realize the periodic table is giving them a real-world average, not a clean textbook integer.
It’s messy in a realistic waylike life, but with more protons.
Finally, electron clues and periodic trends tend to click when you connect them to outcomes you can picture. If sodium tends to lose an electron and
chlorine tends to gain one, you can predict a lot about their bonding without memorizing a giant list. Many learners build confidence here by practicing
small predictions: “Is this element more likely to be a metal or a nonmetal?” “Would it form a +1 ion or a −1 ion?” Those quick questions turn the
periodic table from a static chart into a tool you can actually use.
The best part is that these experiences stack. Once you’ve used the atomic number to confirm an element, used the symbol to avoid a mix-up, used group
position to predict behavior, and used atomic mass to verify details, you stop feeling like you’re guessing. You’re not memorizing a wall chartyou’re
reading it. And that’s when chemistry gets a whole lot less intimidating.