Table of Contents >> Show >> Hide
- What Is Atomic Mass?
- Why Atomic Mass Is Usually a Decimal
- What Are Isotopes?
- The Formula for Calculating Atomic Mass
- Step-by-Step: How to Calculate Atomic Mass
- Example 1: Calculating Atomic Mass of Chlorine
- Example 2: Calculating Atomic Mass of Boron
- Example 3: Finding a Missing Isotope Abundance
- Common Mistakes When Calculating Atomic Mass
- Why Learning Atomic Mass Matters
- Quick Tips for Remembering the Process
- Conclusion: Atomic Mass Is an Average, Not a Mystery
- Experiences and Practical Lessons Related to Learning How to Calculate Atomic Mass in Chemistry
If chemistry had a talent show, atomic mass would be that quiet student who does all the hard math while the flashy elements get the applause. It sits there on the periodic table looking harmless, but once you understand it, a lot of chemistry suddenly starts making sense. Whether you are studying isotopes for the first time, reviewing for an exam, or just trying to decode why chlorine is listed as 35.45 instead of a neat whole number, learning how to calculate atomic mass is one of those skills that pays off again and again.
In simple terms, atomic mass tells you the average mass of an element’s atoms. But there is a catch: most elements exist as a mix of isotopes, which are versions of the same element with different numbers of neutrons. That means the atomic mass on the periodic table is not usually the mass of one exact atom. Instead, it is a weighted average based on the natural abundance of each isotope. Yes, chemistry loves averages almost as much as teachers do.
In this guide, you will learn what atomic mass means, how it differs from mass number, the formula used to calculate it, and several step-by-step examples. By the end, you should be able to look at isotope data and calculate atomic mass without breaking into a dramatic sweat.
What Is Atomic Mass?
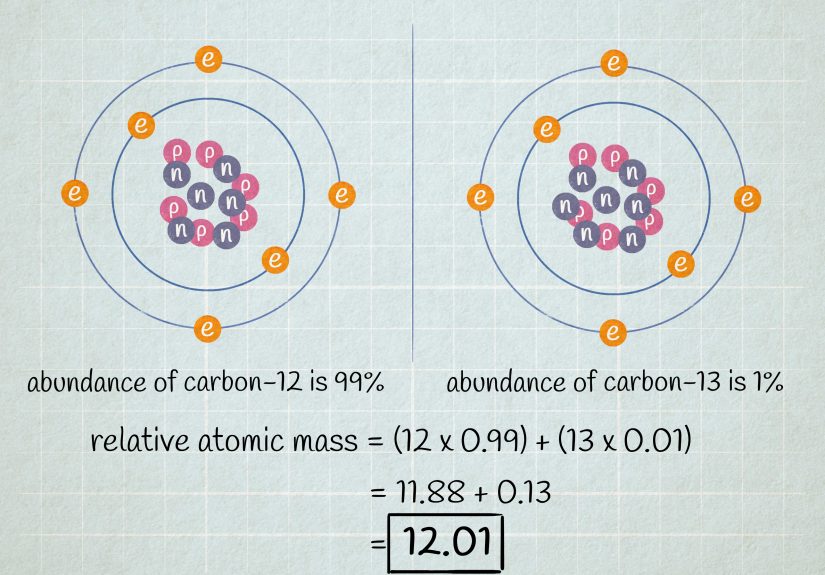
Atomic mass is the weighted average mass of all the naturally occurring isotopes of an element. It is usually expressed in atomic mass units, often abbreviated as amu or simply u. One atomic mass unit is based on carbon-12, which is assigned a value of exactly 12.
That definition matters because it gives chemists a consistent reference point. Instead of measuring atoms with a tiny bathroom scale that does not exist, scientists compare atomic masses to the mass of carbon-12. This allows the periodic table to list values that represent how heavy an element’s atoms are on average.
Atomic Mass vs. Mass Number
Students often confuse atomic mass with mass number, and honestly, chemistry does not help by giving them names that sound like identical twins.
Mass number is the total number of protons and neutrons in one specific atom. It is always a whole number. For example, carbon-12 has a mass number of 12, while carbon-13 has a mass number of 13.
Atomic mass, on the other hand, is the average mass of all naturally occurring isotopes of that element. That is why carbon on the periodic table is listed as about 12.01 rather than exactly 12.
So if mass number is one student’s test score, atomic mass is the class average.
Why Atomic Mass Is Usually a Decimal
If every atom of an element were identical, the atomic mass would be a clean whole number. But nature prefers variety. Many elements have more than one isotope, and those isotopes occur in different percentages. Because the periodic table reflects the average of those isotopes, the result is usually a decimal.
Take chlorine as the classic example. Chlorine exists mostly as chlorine-35 and chlorine-37. Since chlorine-35 is more common, the average atomic mass ends up closer to 35 than to 37. That is why the periodic table shows chlorine at about 35.45.
This decimal is not chemistry trying to be difficult just for fun. It is simply the result of averaging atoms with different masses in the proportions found in nature.
What Are Isotopes?
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Because the number of protons stays the same, the element does not change. Because the number of neutrons changes, the mass changes.
For example, all carbon atoms have 6 protons. But carbon-12 has 6 neutrons, while carbon-13 has 7 neutrons. Same element, different mass. That is the isotope story in one sentence.
Isotopes matter in chemistry because they affect atomic mass. They also show up in medicine, dating fossils, environmental science, and lab analysis. So while they may seem like a small textbook detail, isotopes are actually doing quite a lot behind the scenes.
The Formula for Calculating Atomic Mass
To calculate atomic mass, you use a weighted average formula:
Atomic Mass = (Isotope 1 mass × isotope 1 abundance) + (Isotope 2 mass × isotope 2 abundance) + …
There are two important rules here:
1. Convert percent abundance to decimal form
If an isotope is 75% abundant, use 0.75 in the calculation, not 75.
2. Multiply first, then add
You multiply each isotope’s mass by its decimal abundance, then add the products together.
That is it. No smoke, no mirrors, no chemistry wizard robe required.
Step-by-Step: How to Calculate Atomic Mass
Let’s break the process into a method you can repeat on homework, quizzes, and exams.
Step 1: List the isotopes and their masses
Write down each isotope involved and its atomic mass.
Step 2: Write the natural abundance of each isotope
This is usually given as a percentage.
Step 3: Convert each percentage to a decimal
Divide by 100. For example, 24.3% becomes 0.243.
Step 4: Multiply each isotope’s mass by its decimal abundance
This gives the weighted contribution of that isotope.
Step 5: Add the weighted values together
The sum is the atomic mass of the element.
Once you do this a few times, it starts to feel routine. At first it looks like serious chemistry. After a while, it is basically just careful arithmetic wearing safety goggles.
Example 1: Calculating Atomic Mass of Chlorine
Suppose chlorine has the following isotopes:
- Chlorine-35: mass = 34.97 amu, abundance = 75.77%
- Chlorine-37: mass = 36.97 amu, abundance = 24.23%
Convert percentages to decimals
- 75.77% = 0.7577
- 24.23% = 0.2423
Multiply mass by abundance
- 34.97 × 0.7577 = 26.50
- 36.97 × 0.2423 = 8.96
Add the results
26.50 + 8.96 = 35.46 amu
So the atomic mass of chlorine is approximately 35.46 amu. This matches the periodic table value very closely.
Notice how the answer is closer to 35 than 37. That is because chlorine-35 is more abundant. Weighted averages always lean toward the value that appears more often.
Example 2: Calculating Atomic Mass of Boron
Now let’s try boron, another favorite in chemistry class.
- Boron-10: mass = 10.01 amu, abundance = 19.9%
- Boron-11: mass = 11.01 amu, abundance = 80.1%
Convert to decimals
- 19.9% = 0.199
- 80.1% = 0.801
Multiply
- 10.01 × 0.199 = 1.99
- 11.01 × 0.801 = 8.82
Add
1.99 + 8.82 = 10.81 amu
The atomic mass of boron is about 10.81 amu. Again, the answer is closer to the isotope that is more common, which is boron-11.
Example 3: Finding a Missing Isotope Abundance
Sometimes chemistry problems get sneaky and ask you to find the abundance of an isotope instead of calculating the final atomic mass directly.
Imagine an element has two isotopes:
- Isotope A: mass = 20.0 amu
- Isotope B: mass = 22.0 amu
The average atomic mass is 20.4 amu. What is the abundance of isotope A?
Let the abundance of isotope A be x. Then the abundance of isotope B must be 1 – x.
Set up the equation:
(20.0 × x) + (22.0 × (1 – x)) = 20.4
Now solve it:
20x + 22 – 22x = 20.4
-2x + 22 = 20.4
-2x = -1.6
x = 0.8
So isotope A has an abundance of 80%, and isotope B has an abundance of 20%.
This type of problem looks scarier than it is. It is still the same weighted average idea, just with algebra invited to the party.
Common Mistakes When Calculating Atomic Mass
Forgetting to convert percentages into decimals
This is probably the most common error. If you use 75 instead of 0.75, your answer will launch itself into another galaxy.
Using mass number instead of isotope mass
Mass numbers are whole numbers, but actual isotope masses may include decimals. In many classroom problems, the distinction is small, but using the correct isotope mass gives a more accurate answer.
Not checking whether abundances add to 100%
If the isotope percentages do not total 100%, something is off. Double-check the data before calculating.
Assuming atomic mass equals the most common isotope exactly
The most abundant isotope has the biggest influence, but the average is still an average. Close does not mean identical.
Why Learning Atomic Mass Matters
Atomic mass is not just a chapter test topic. It is a foundation for bigger chemistry ideas. You use atomic mass to calculate molar mass, predict masses in chemical reactions, understand isotopic notation, and interpret data from instruments such as mass spectrometers.
It also trains you to think like a scientist. Instead of asking, “What is one atom like?” you ask, “What does a real sample look like overall?” That shift from individual particles to measurable averages is a huge part of chemistry.
In short, atomic mass helps connect the tiny atomic world to the practical calculations chemists perform in labs, classrooms, and industry.
Quick Tips for Remembering the Process
- Identify the isotopes
- Decimalize the abundances
- Multiply mass by abundance
- Add the products
You can remember it as IDMA. It is not the most glamorous acronym in science history, but it works.
Conclusion: Atomic Mass Is an Average, Not a Mystery
Once you understand isotopes, atomic mass becomes far less intimidating. The key idea is simple: the atomic mass of an element is the weighted average of its naturally occurring isotopes. That means you multiply each isotope’s mass by its abundance, add the results, and end up with the average value shown on the periodic table.
Whether you are calculating chlorine’s mass, solving for a missing isotope abundance, or trying to make sense of why the periodic table loves decimals so much, the same logic applies every time. Learn the pattern, practice a few examples, and atomic mass goes from confusing to surprisingly manageable.
And that is the beauty of chemistry. What first looks like a weird decimal on a chart turns out to be a story about nature, probability, and averages. Not bad for a number that spends most of its life sitting quietly under an element symbol.
Experiences and Practical Lessons Related to Learning How to Calculate Atomic Mass in Chemistry
For many students, the first experience with atomic mass is not exactly magical. It usually arrives in the middle of a unit on atoms, isotopes, and the periodic table, right around the time everyone is still trying to remember whether neutrons are neutral or just emotionally unavailable. At first glance, atomic mass can feel unnecessarily complicated. Students see whole-number isotope names like oxygen-16 and neon-20, then look at the periodic table and find values like 15.999 or 20.18. The immediate reaction is often, “Chemistry, are you okay?”
But something interesting happens once students work through a few actual problems. The topic starts to click because it connects math to a real scientific reason. In class settings, one of the most helpful experiences is using colored candies, beads, or coins to represent isotopes. A teacher might say that red candies are one isotope and blue candies are another, each with a different “mass.” Once students count how many of each color are in a sample and calculate the weighted average, atomic mass suddenly feels less abstract. It becomes clear that the periodic table is showing an average based on what is actually found in nature, not the mass of one random atom picked out of a microscopic crowd.
Another common learning experience comes from making mistakes, which is chemistry’s unofficial teaching assistant. Many students forget to convert percentages into decimals the first time they calculate atomic mass. The answer then comes out absurdly large, which is actually useful because it teaches an important habit: always sanity-check the result. If the isotopes have masses around 35 and 37, your final answer should not be 3,546. That moment of catching the mistake often sticks longer than the correct answer itself.
Students also tend to remember atomic mass better when it is linked to real-world tools like mass spectrometry. Even a basic explanation that scientists can separate isotopes and measure their relative abundances helps the concept feel grounded in reality. It is not just textbook math invented to torment sleepy teenagers before lunch. It reflects how scientists analyze elements, verify isotopic composition, and study materials in research, medicine, and environmental science.
From a study-skills perspective, the best experience is repeated, low-stress practice. Atomic mass problems become much easier when students stop treating each one like a brand-new puzzle and start seeing the same pattern every time: write the isotopes, convert the percentages, multiply, then add. Once that rhythm develops, confidence grows fast. Many learners go from “I do not understand this at all” to “Oh, this is just a weighted average” in a single homework session.
In the end, learning how to calculate atomic mass is a classic chemistry experience: confusing at first, logical once explained, and oddly satisfying when the numbers work out. It teaches more than one skill. It builds precision, reinforces scientific reasoning, and shows how averages can describe the real world at the atomic level. That is a pretty solid payoff for a topic that begins with a decimal and a mild sense of betrayal.





